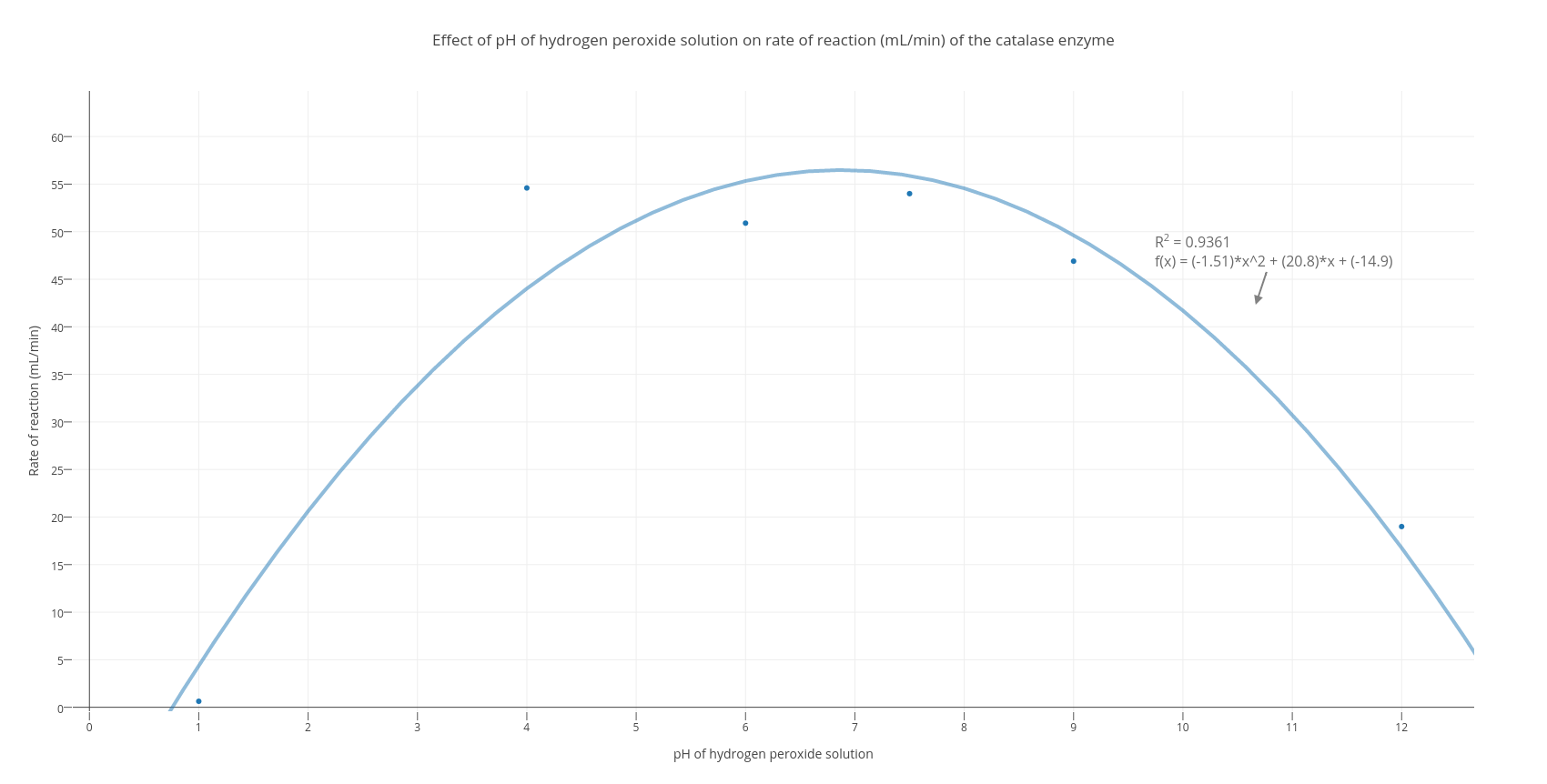
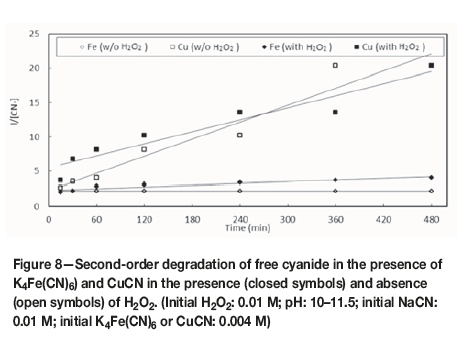
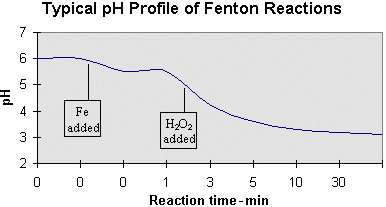
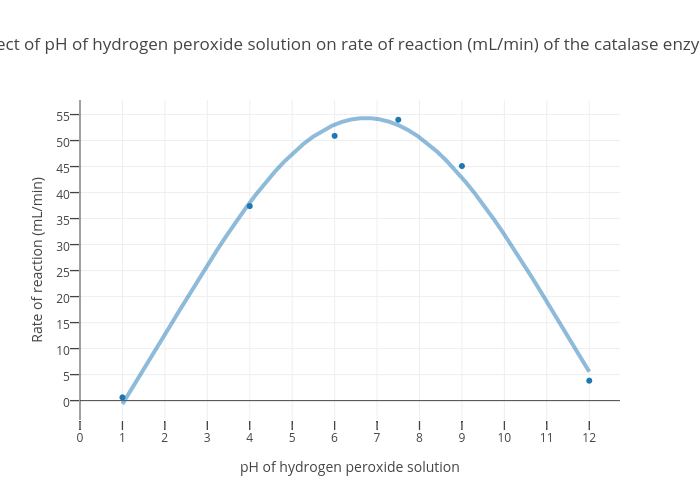
Effect of pH on DR removal; hydrogen peroxide = 75mg/L , irradiation... | Download Scientific Diagram

Aqueous decomposition behavior of solid peroxides: Effect of pH and buffer composition on oxygen and hydrogen peroxide formation - ScienceDirect

The life story of hydrogen peroxide II: a periodic pH and thermochemical drive for the RNA world | Journal of The Royal Society Interface

Frozen Hydrogen Peroxide and Nitrite Solution: The Acceleration of Benzoic Acid Oxidation via the Decreased pH in Ice | Environmental Science & Technology

The life story of hydrogen peroxide II: a periodic pH and thermochemical drive for the RNA world | Journal of The Royal Society Interface
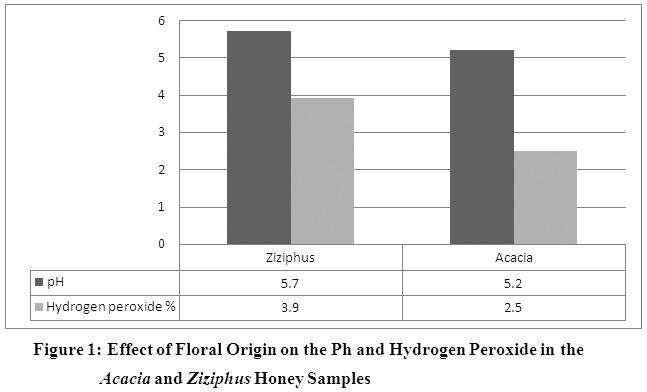
Hydrogen Peroxide and Dicarbonyl Compounds Concentration in Honey Samples from Different Botanical Origins and Altitudes in the South of Saudi Arabia
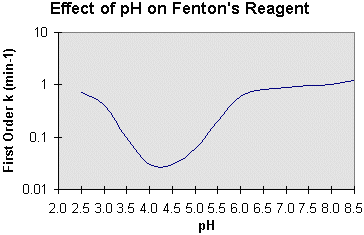
![Effect of pH on the decomposition of hydrogen peroxide ([H 2 O 2 ] 0 :... | Download Scientific Diagram Effect of pH on the decomposition of hydrogen peroxide ([H 2 O 2 ] 0 :... | Download Scientific Diagram](https://www.researchgate.net/profile/Haci-Deveci/publication/234110563/figure/fig2/AS:667856089911306@1536240708857/Effect-of-initial-concentration-of-H-2-O-2-on-the-decomposition-of-hydrogen-peroxide-pH_Q320.jpg)
Effect of pH on the decomposition of hydrogen peroxide ([H 2 O 2 ] 0 :... | Download Scientific Diagram