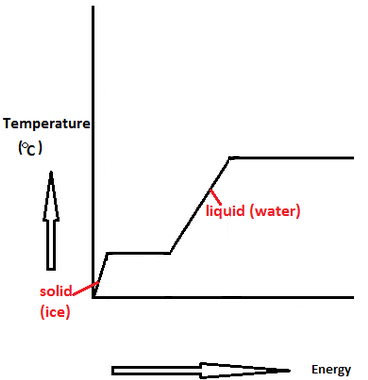
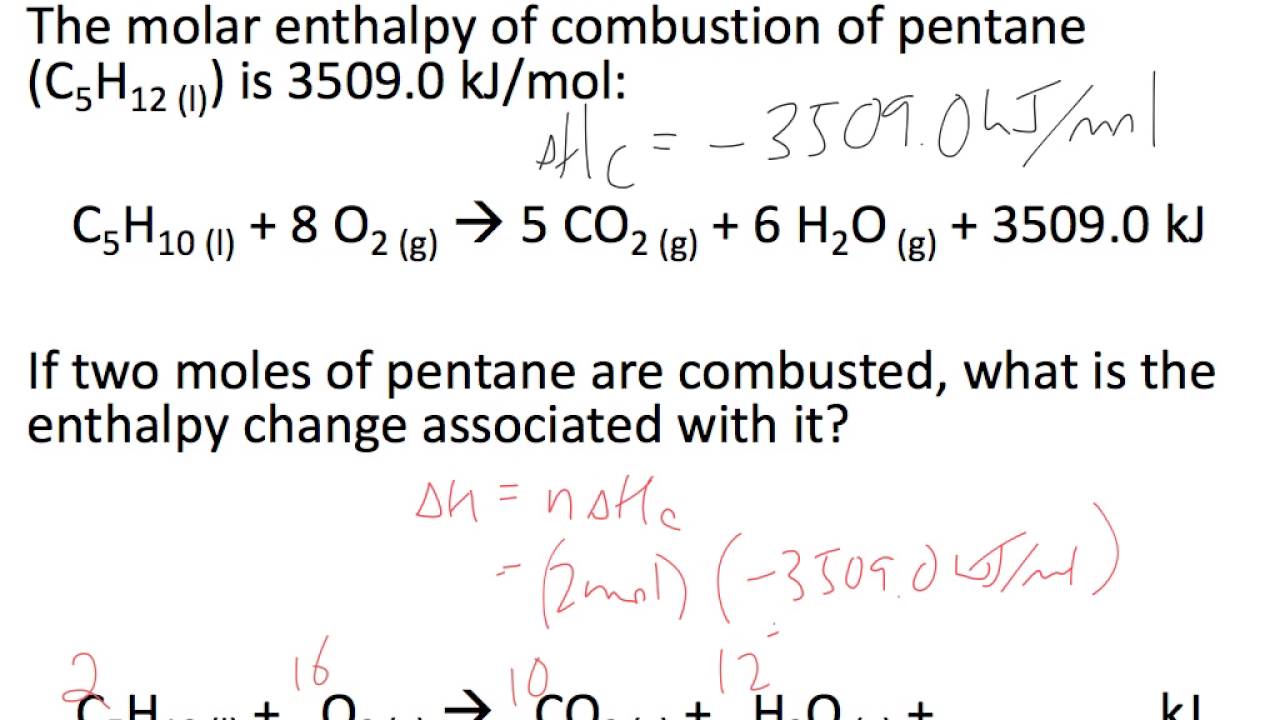Calculate the change in entropy for the fusion of 1 mol of ice. The melting point of ice is 300K and molar enthalpy of fustion for ice = 6.0 k J mol^(-1).

Calculations in Chapter 10. Molar Enthalpy of Fusion Used when melting or freezing = ___energy ____ mol of substance Can be arranged to find any of the. - ppt download

Heat of Fusion Equation & Examples | What is Heat of Fusion? - Video & Lesson Transcript | Study.com

Specific Heat, J/g.°C 2.06 - ice 4.18 - water 2.03 - steam.Molar heat of fusion for water, kJ/mol - 6.02 - Home Work Help - Learn CBSE Forum

Calculate the change in entropy for the fusion of 1 mole of ice (water). The melting point of water - YouTube

Specific Heat, J/g.°C 2.06 - ice 4.18 - water 2.03 - steam.Molar heat of fusion for water, kJ/mol - 6.02 - Home Work Help - Learn CBSE Forum

Using Heat of Fusion or Vaporization to Find the Heat Needed to Melt or Boil a Substance | Chemistry | Study.com