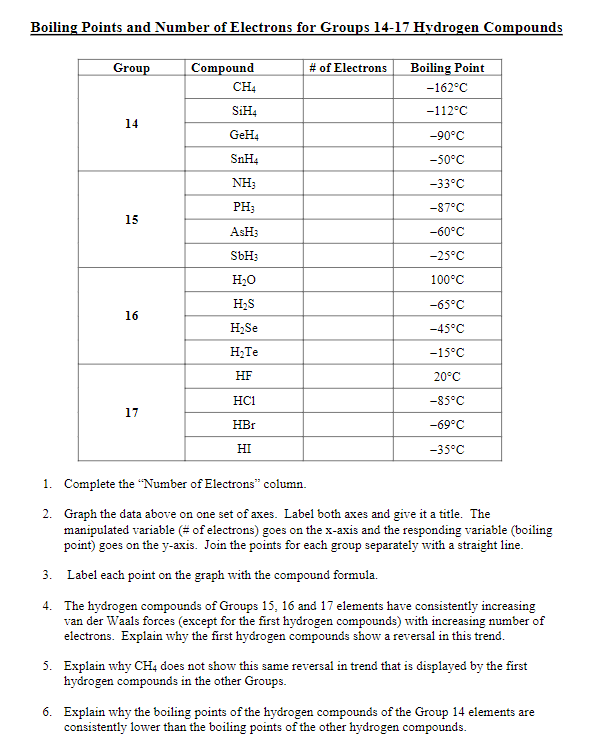
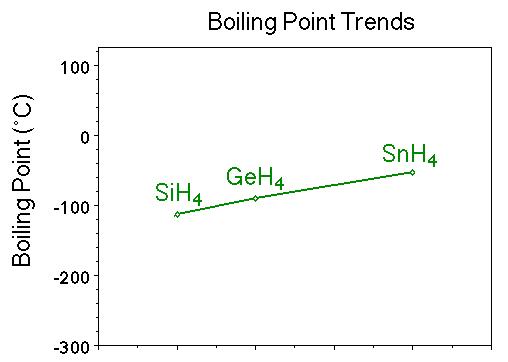
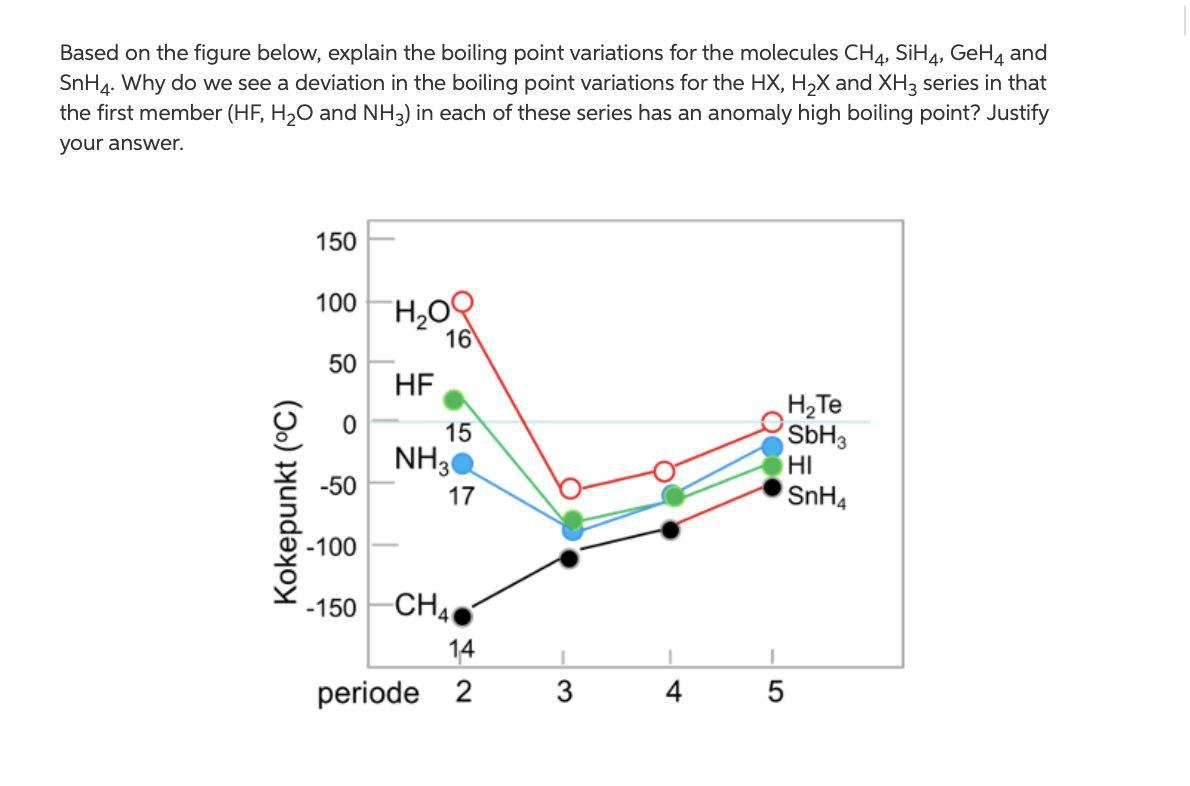
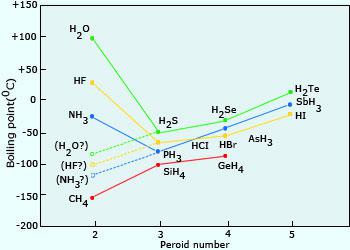
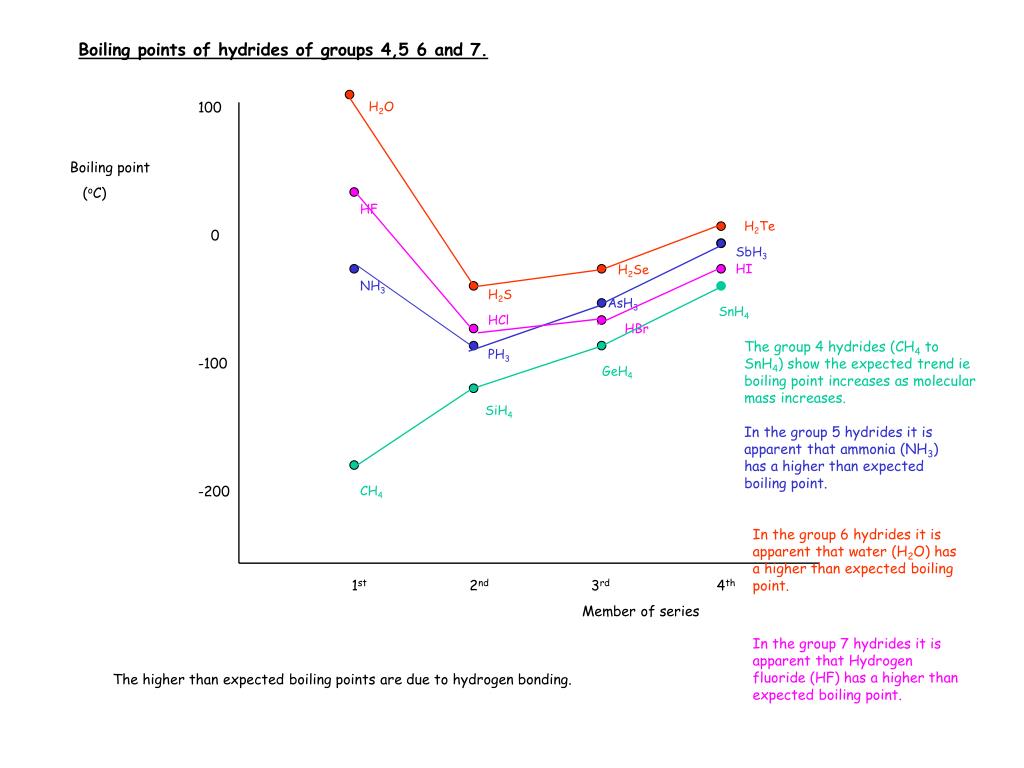
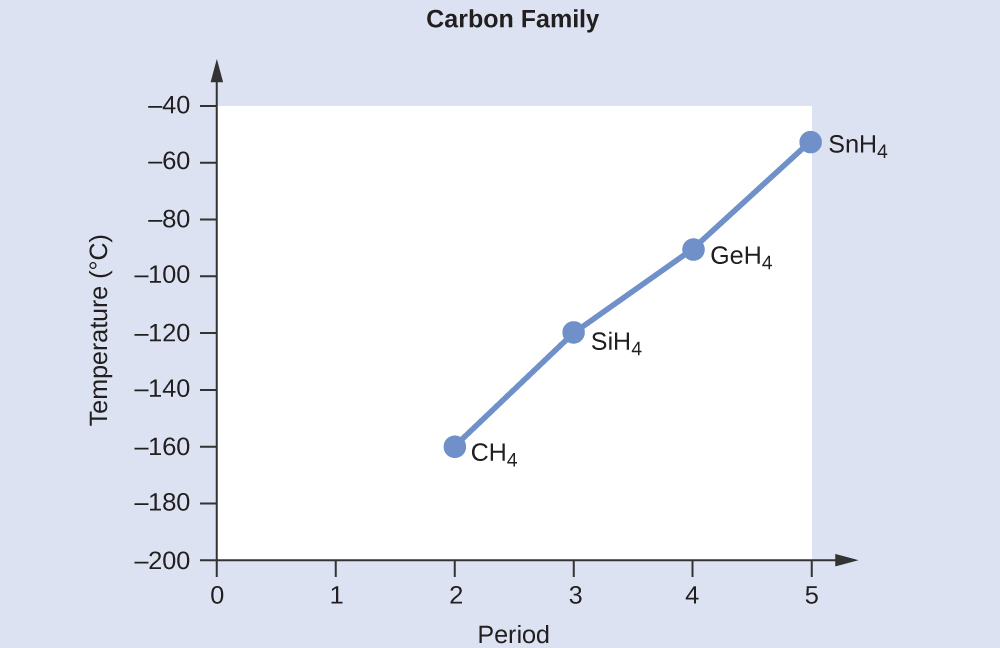
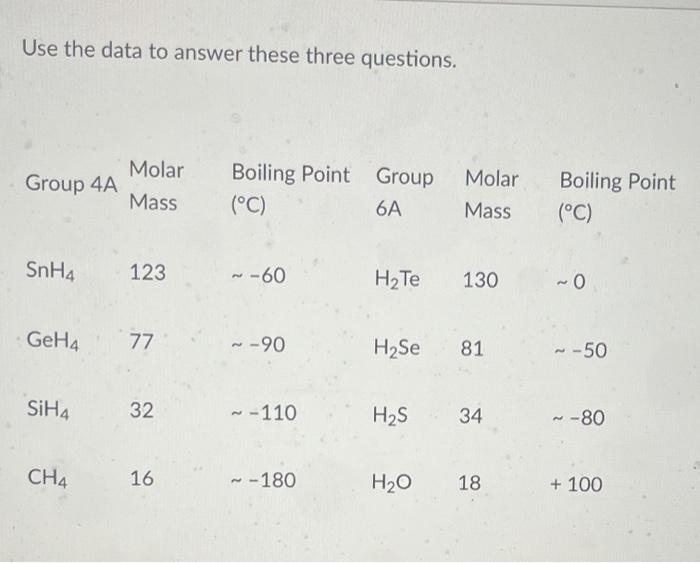
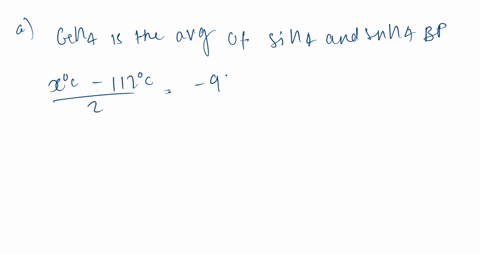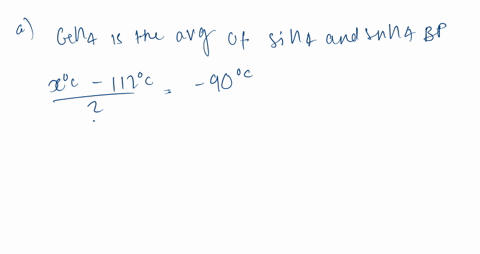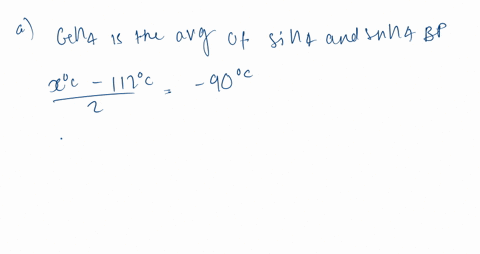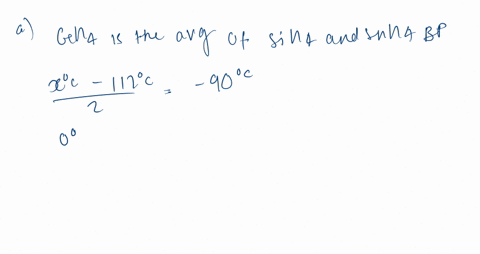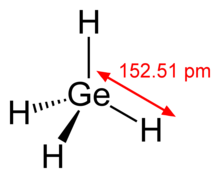Consider the following: CH4, SiH4, GeH4, SnH4 The boiling points for these compounds increase roughly at the same rate except for CH4. Why does CH4 have a significantly lower boiling point than
Consider the following: CH4, SiH4, GeH4, SnH4 The boiling points for these compounds increase roughly at the same rate except for CH4. Why does CH4 have a significantly lower boiling point than