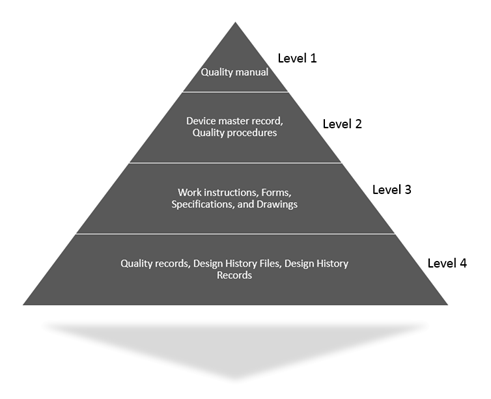
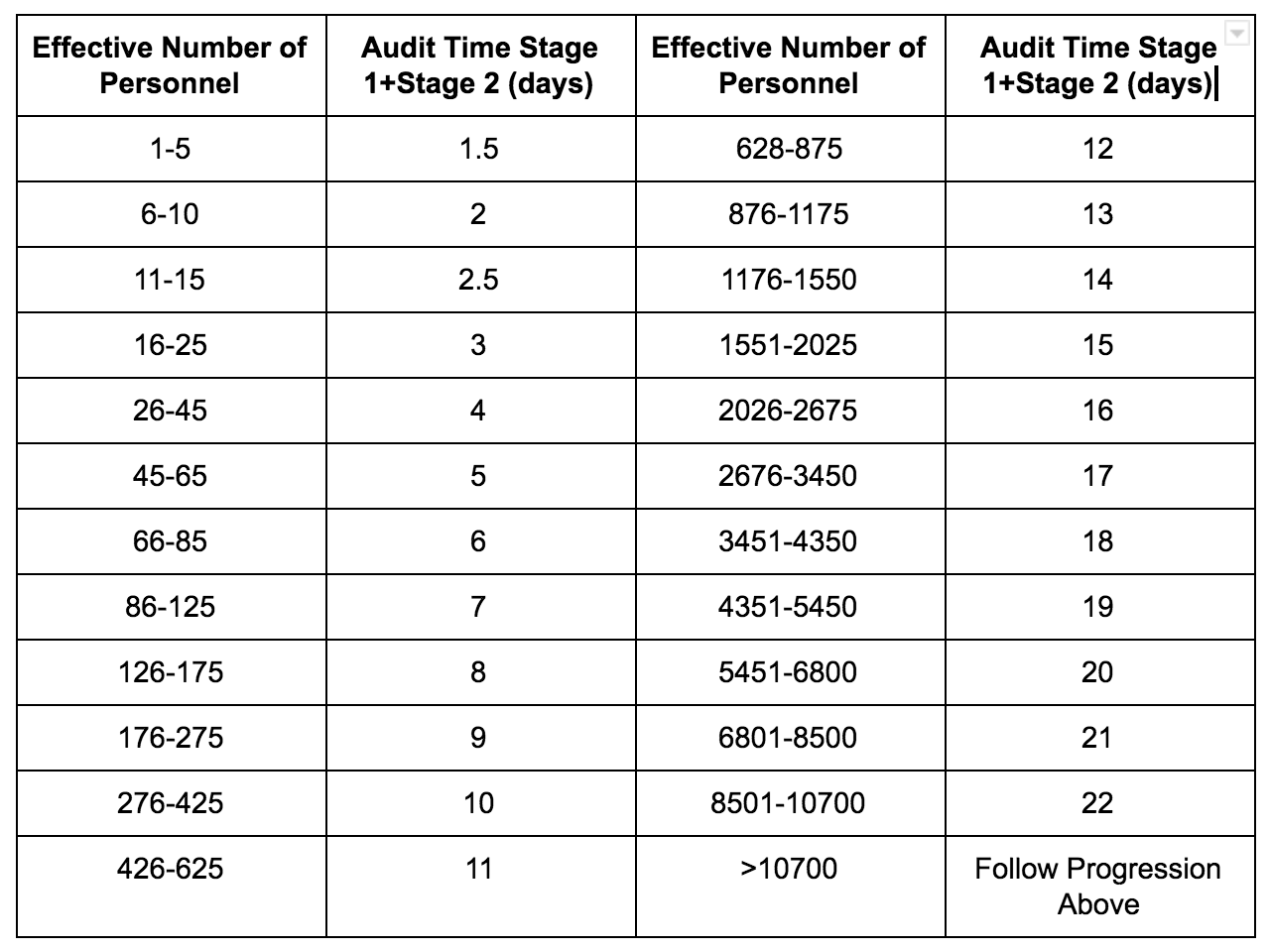
Poor Quality of ISO Documents | Cost of Quality Calculation Example | ISO 9001 Quality System - YouTube

ISO 13485 – Medical Device Quality Management System Requirements – ISO Templates and Documents Download

Amazon.com: ISO 13485 - Obstacle or Opportunity: How to live a QM system to benefit companies and employees eBook : Kalchschmid-Lehmann, Andreas: Kindle Store